|
An ion is an atom or molecule with a net electric charge due to the loss or gain of one or more electrons.
|
Ionic bonding is the complete transfer of valence electrons between atoms. Ions bond to achieve Nuclear Stability By sharing valence electrons
|
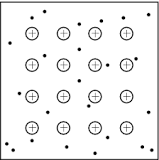
Metallic Bonding is when there are electrons that are not shared so they stay around positive ions and act like a "glue" to give the substance a more definite structure.
Ionic compound Bonds
Step 1: Write the symbols of the ions with the oxidation number
|
Example:
|
Rules for naming Ionic Compounds
|
Step 1. The positive ion is written first in the name then the negative ion is written second in the name.
Step 2. The name of the positive ion is the same as the element from which it is derived Step 3. The negative ion is named by adding the suffix -ide to the root of the element name |
Example:
Sodium + Oxygen Na+2 O-1 Sodium oxide |
